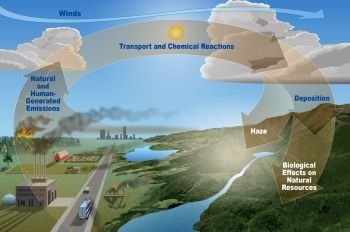
Acid Rain
Rain or snow that contains significant amounts of sulfuric acid or nitric acid. Sulfuric or nitric acid is formed when sulfur dioxide (SO,) or nitrogen oxide (NO,) emitted by industry or transport undergo a chemical transformation in the atmosphere.
The US National Commission on Air Quality, in its 1981 report, found that the process by which manmade pollutants were transformed into acid rain was now ‘reasonably well known’. It described how SO2 or NO, gas released into the atmosphere is first oxidized to sulfate or nitrate particles. If water vapor is present, the particles are further transformed into sulfuric or nitric acid which contaminates rain, snow, or fog. Alternatively, sulfate or nitrate particulates may be deposited on the ground in dry form, later combining with surface water or groundwater to produce acid.
In the United States, rain that is ten to 40 times as acidic as normal rainfall has been occurring frequently in many parts of New England and New York. The average pH of rainfall over substantial portions of the Northeast is 4.3, approximately ten times normal acidity. Comparable levels of acidity are occurring in many parts of Canada. Highly acidic rainfall has also been observed in a number of southeastern states, particularly Florida, Virginia, Texas, and North Carolina, in the Northern Plains states of Minnesota and Wisconsin, and in the Colorado Rockies.
In 1979, the US President established a ten-year comprehensive Federal Acid Rain Assessment Program to be planned and managed by a standing Acid Rain Co-ordination Committee. The assessment program includes applied and basic research on acid rain effects, monitoring, transport modes, and the study of control measures. Collaboration with the Canadian and Mexican governments, and with other nations and international organizations, was anticipated.
In August, 1984, New York became the first state to legislate on acid rain. By the end of that year, the areas of the state most at risk had to be identified, and tolerance levels for sulfur deposition determined. Rules governing emissions from the industrial burning of coal and oil were to be produced by 1986, and to come into force by 1988.
With respect to the acid rain issue, the debate in the US Congress has split along regional lines: members from the Northeast have pressed for a tough control program, while members from the Midwest fear a control program could hamper the region’s economy. The West, neither a primary victim of nor contributor to acid rain does not want to pay to control it.
The Swedish Fisheries Board has now done a national survey of Swedish lakes and has found that 18 000 out of a total of about 90 to 100 thousand are acid. These lakes, poor in calcium, magnesium and nutrients, are situated (mainly in the southwest of Sweden) in areas with coniferous forests and pre-Cambrian bedrock. The pH of the acid lakes varies between four and about Lakes have natural defense mechanisms or buffering capacity caused by the presence of hydrogen carbonate. However, the majority of Scandinavian lakes and watercourses has low lime content, and thus a low content of hydrogen carbonate, and are therefore particularly vulnerable to acidification.
While it was agreed that there were many factors involved in the acidification of lakes and soils in Sweden and other parts of Scandinavia, there was no doubt that sulfur and nitrogen compounds resulting from combustion of fossil fuels in Europe were transported over long distances and contributed to the acidification problem in vulnerable areas.
Acid precipitation over southern Norway has increased considerably in recent years. Measurements have shown that there is a marked decrease in the pH value in rivers and lakes after rain. In winter, grey snow has sometimes been observed. In the southernmost counties in particular the soil is poor with low calcium levels, so that acid precipitation is only partly neutralized by the soil and thus immediately affects the pH value of rivers and lakes. An observed decline in stocks of fish in a large number of lakes may be related to
increased acidity. Acid also has a leaching effect on mineral substances in the soil with possible detrimental effects on forest growth. Norway takes considerable interest in the new OECD research project on the long-range movement of air pollutants.
An October 1983 report, to the West German Bundestag (federal parliament) suggested that more than one-third of West German forests had been damaged by acid rain. In Bavaria and the Black Forest it was claimed that nearly half of the trees had been damaged. New limits on emissions from power stations were announced later in 1983 aiming to reduce SO2 emissions by over 85 per cent.
In 1984, the Netherlands Environment Minister released a plan to reduce industrial SO2 emissions by 70 per cent, NO, emissions from motor vehicles by 30 per cent and ammonia released from agricultural fertilizers by 50 per cent, by the end of the century. During the same year, the French government announced its intentions to halve SO2 emissions by 1990.
However, the United States and British governments continued to question the relationships between SO2 emissions and acid deposition. The US Clean Air Act of 1990 aimed at cutting SO2 emissions virtually in half by the year 2000. This was Phase 1 of the US Acid Rain Program linked with the US Emission Trading Program. Phase 2 begins in 2000 with a further substantial reduction in sulfur emissions.
In 1998, Resources for the Future published the first integrated assessment of both the projected costs and benefits of Title IV of the 1990 amendments to the US Clean Air Act. The report concluded that the dollar value of reduced mortality risk alone measured several times the expected costs of compliance with the acid rain program.