VIDEO
Carbon Dating: (How) Does It Work?
Carbon Dating
Isotopes are also used in the dating of ancient soils, plants, animals, and the tools of early peoples. An isotope of carbon, 14C, which has a half-life of 5730 years, can be used to calculate geological age. Since the radioactive decay rate of carbon is constant, observing its decay rate allows the measurement of the number of years that have past compared to carbon’s half-life.
The preservation of the original organic sample can affect carbon dating. Carbon-14, which decays to 14N, is mostly used for dating samples of fairly recent geological age. Most scientists believe that carbon dating is only accurate for dating specimens thought to be between 30,000 and 50,000 years old. Carbon dating is particularly helpful when finding the age of bone, wood, shell, fossils, and other organic samples since they all contain carbon.
These plants and animals use carbon in their basic structure and usually have a good amount of carbon left to be measured. Other radiometric methods that make use of uranium, lead, potassium, and argon measure much longer time periods since they are not limited to the organic remains of prehistoric samples.
The table bellow shows a few of the elements used to date samples of different ages. Dating samples is not an exact science. A lot of factors have to be considered when a sample is dated, including its preservation and the amount of erosion and exposure it has suffered. For example, when lead is depleted from a rock sample through erosion, then the uranium used to date the sample through the breakdown of the uranium to the lead end product (daughter) would show an incorrectly young age.
|
Isotopes (original) – (range in years) |
Isotopes (decay products)
|
Half-life (years)
|
Dating accuracy (range in years) |
|
Carbon-14 |
Nitrogen-14 |
5730 |
30,000–60,000 |
|
Rubidium-87 |
Strontium-87 |
4.8 billion |
10 million–4.6 billion |
|
Potassium-40 |
Argon-40 |
1.3 billion |
50,000–4.6 billion |
|
Uranium-238 |
Lead-206 |
4.5 billion |
10 million–4.6 billion |
Geochronological Units
If you visit a Museum of Natural Sciences or Natural History you won’t be looking at brightly colored paintings or finely crafted statues created by human artists. Instead, you will see thousands of fossils and preserved shells and bones of ancient marine life, plants, and animals created by Nature.
These exhibits provide a chronological history of the Earth by displaying rock, plant, and animal specimens. In addition, besides giving the location where each sample was discovered, most museums also date samples according to their place in geological time.
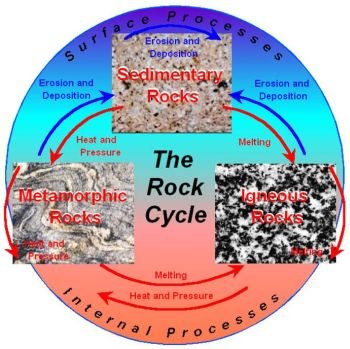
Geological time is measured and divided into various parts called geochronological units.
Although people sometimes use the word eon to mean a really long time, like ‘‘it has been eons since I visited with my cousin,’’ the term actually comes from the Greek word, eos, meaning ‘‘dawn.’’ Geochronological units start with the major divisions of time called eons. Eons are measured in millions of years. As time dating is refined, the boundaries of the eons may change slightly, but most geological dating is calculated with a margin of error +/-60 million years either way.
The three major eon divisions are the Archean, Proterozoic, and Phanerozoic. The Archean (Greek for ‘‘ancient’’) eon is commonly thought to include the oldest rocks known. It is often called the early Precambrian era which begins with the formation of the Earth, about four billion years ago until about 2500 million years ago. The Proterozoic eon is now thought of as being part of the late Precambrian era.
.