Pollution Greenhouse Effect (GCSE Chemistry)
Greenhouse Chemistry
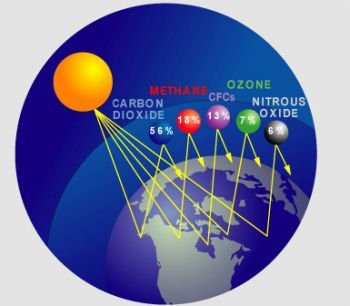
The human presence on the earth has developed to the point where we have acquired the ability affect the earth’s climate. At one time, the gases our historical ancestors put into the atmosphere by burning wood to cook the animals they hunted was insignificant. The small quantities of greenhouse gases they produced were absorbed and diluted by the air. Today, this is no longer the case. These texts will introduce you to the greenhouse gases-where they come from, where they go, and how they absorb heat. Some of the components of the atmosphere are natural. Some are natural substances at unnatural concentrations. Some are exclusively a human contribution. Since all fuels-with the single exception of hydrogen gas-have carbon as part of their chemical makeup, every fuel produces carbon dioxide when it burns.
Gases in the Atmosphere
The following gases make up our atmosphere:
Nitrogen, 78 percent
Oxygen, 21 percent
Water vapor, 0–2 percent
Argon, 0.1 percent
Carbon dioxide, 0.04 percent [280 parts per million (ppm) preindustrial, 379 ppm in 2005]
Methane, 0.0002 percent [715 parts per billion (ppb) pre-industrial, 1774 ppb in 2005]
Nitrogen dioxide, 0.00003 percent (270 ppb preindustrial, 319 ppb in 2005)
Stratospheric ozone, trace amount
Tropospheric ozone, trace amount
Generating Carbon Dioxide
THE CARBON CYCLE
Carbon dioxide enters the atmosphere naturally by
• Anaerobic bacteria that decompose other organic matter
• Animals that exhale carbon dioxide during respiration
• Occasional volcanic activity
Nature removes carbon dioxide from the atmosphere by
• Plants consuming it in the process of photosynthesis
• Water dissolving carbon dioxide
Some of the carbon dioxide that is released into the air is removed by being dissolved in the oceans and other bodies of water. The amount of carbon dioxide that can dissolve in a volume of water depends on the temperature of the water and the concentration of carbon dioxide that is already dissolved in the water. Warmer water dissolves less carbon dioxide than cooler water. Water with a higher concentration of carbon dioxide also dissolves less carbon dioxide than water with little carbon dioxide. This process is self-limiting because as the greenhouse effect causes higher temperatures, the removal rate of the carbon dioxide decreases.
BURNING FOSSIL FUELS
The fossil fuels are coal, oil, and natural gas. The name fossil fuels derive from the origin of these fuels from the remains of organic matter preserved from prehistoric times. Most of our energy used for electricity and transportation today comes from the burning of fossil fuels, which generates carbon dioxide as a by-product. As a result of fossil fuel combustion, carbon dioxide in the atmosphere is now 35 percent higher than it was a century and a half ago.
Whenever anything containing carbon burns, a product of that combustion is carbon dioxide and water vapor. The basic chemical reaction for the complete combustion of a fuel is
Fuel + oxygen (O2) → carbon dioxide (CO2) + water (H2O)
In this equation, the arrow stands for produces or yields. Sometimes the combustion is not complete, which results in production of carbon monoxide and only partial breakup of the fuel molecules.
Notice that every one of these fuels except hydrogen contains carbon. If there is a C in the formula, it contains carbon. (H represents a hydrogen atom, and O represents an oxygen atom.) Carbon dioxide has the formula CO2, which means that it has one carbon atom and two oxygenatoms. Oxygen in the atmosphere has the formula O2. This means that it has two oxygen atoms that make up the gas oxygen that is a constituent of the atmosphere. The atoms you start with get rearranged in any chemical reaction to form new substances with properties that are different from those of the substances you started out with.
All fuels (except hydrogen) have carbon in their chemical formula and produce carbon dioxide when they burn.
CEMENT PRODUCTION-PAVING PARADISE
Humans put carbon dioxide into the atmosphere in other ways besides burning fossil fuels. One example is the production of concrete, which involves the conversion of calcium carbonate rock (limestone) into calcium oxide. This process also produces carbon dioxide, as shown in this reaction:
Calcium carbonate + oxygen + heat → calcium oxide + carbon dioxide
Development of cities and towns and industries contributes to global warming in at least three ways:
1. It releases carbon dioxide directly to the air through the production of cement.
2. It requires heat, which is likely to involve the combustion of fossil fuels.
3. It often replaces vegetation that absorbs carbon dioxide from the atmosphere.
CEMENT MANUFACTURING
Carbon dioxide is released when limestone is heated to produce quicklime during the production of cement.
Absorption of Light by Gases in the Atmosphere
Fossil fuels are a concern because they increase the absorption of incoming solar radiation above the amount absorbed naturally. This is referred to as the enhanced greenhouse effect.
When a gas molecule absorbs energy, it has no place to store that energy other than in some form of movement of the molecule itself. According to the kinetic theory of matter, molecules are constantly in motion. The molecules move faster when their temperature goes up. Gas molecules in the atmosphere can absorb energy passing through the atmosphere in the form of visible light (from the sun) or invisible electromagnetic waves (from the earth). Some molecules are able to move in more ways than others, enabling them to be better absorbers of certain kinds of light (or electromagnetic) energy.
If we look at the chemical structure of some of the gases in the atmosphere, we can understand better how they absorb light energy passing through the atmosphere.
Let’s start with nitrogen. Nitrogen gas molecules each have two atoms of the element nitrogen bonded together to form a molecule of nitrogen gas that has the formula N2 (which means two nitrogen atoms).
Because the nitrogen gas molecule is so simple, it cannot do very much with the light energy that it absorbs. It can spin or vibrate only a little bit by stretching and pulling.
Because of their structural simplicity, nitrogen and oxygen, which together make up 99 percent of the earth’s atmosphere, absorb relatively small amounts of the visible light energy coming from the sun that passes through the air. This leaves the “glass” of the earth’s greenhouse pretty clear.
Water vapor, however, is a different story. Water (H2O) has two hydrogen atoms bonded to a single oxygen atom. The oxygen atoms are bent to form a 105-degree angle.
The water molecule can twist, turn, gyrate, bend, fl ex, and do its own little chemical dance. Because water has more ways to move, it is better able to absorb light energy.
Water vapor in the atmosphere absorbs certain colors of light (visible wavelengths) on the way down to the earth’s surface as if there were a fi lm on the glass of earth’s greenhouse causing it to be less than perfectly transparent. Water also absorbs some of the heat energy in the form of infrared light (long invisible wavelengths) emitted by the earth. For this reason, water is a natural greenhouse gas and contributes to the absorption of heat in the atmosphere. However, as a consequence of global warming, there is more water vapor in the atmosphere, and this, in turn, absorbs even more heat.
The carbon dioxide molecule, like the water molecule, has three atoms. These atoms, rather than being bent, are arranged in a straight line. Because of its molecular structure, carbon dioxide happens to be especially well suited to absorb certain wavelengths (mostly the invisible infrared radiation emitted by the earth). It does not strongly absorb the light energy coming from the sun.
Carbon dioxide lets sunlight through the atmosphere but does not let the energy emitted by the earth pass back into space. In this way, carbon dioxide acts a greenhouse gas.
Methane has four hydrogen atoms bonded to a central carbon atom. Looking at the structure of the methane molecule, one might (correctly) guess that it has a number of ways to vibrate as it absorbs light (and other invisible electromagnetic) waves. Like a good dancer, methane has “lots of moves.” For this reason, methane is an “energy sponge” that absorbs sunlight 20 times more strongly than carbon dioxide.





