Environmental Issues in Atmospheric Chemistry
Atmospheric Chemistry
Since the beginning of industrial development and its associated increase in the burning of fossil fuels, humans have been altering the composition and chemistry of the atmosphere. More recently, the introduction of human made halocarbons has also had an impact on atmospheric composition. The atmosphere is a mixture of gases that includes about 78% nitrogen and 21% oxygen at Earth’s surface. The rest of the atmosphere near Earth’s surface is made up of carbon dioxide and trace amounts of a number of other gases. It is the increase in carbon dioxide and trace gases that has altered atmospheric chemistry.
Historical Background and Scientific Foundations
The carbon dioxide content of the atmosphere has grown significantly in the last 50 years as a byproduct of the burning of fossil fuels. There has been a 36% increase in atmospheric carbon dioxide since the mid-1800s with almost all of the increase due to human activities.
Methane, a trace gas, is naturally found in the atmosphere as the product of decaying organic matter. The concentration of methane in the atmosphere has risen to 148% above its pre-industrial age level. Anthropogenic (human-caused) sources of methane include landfills, the management of livestock, and natural gas and petroleum processing.
Nitrous oxide is another gas that is naturally found in trace amounts in the atmosphere. The concentration of nitrous oxide is largely derived from agricultural sources, with vehicle emissions also contributing nitrous oxide to the atmosphere. The concentration of nitrous oxide in the atmosphere has risen approximately 18% in the last 200 years.
Carbon dioxide, methane, and nitrous oxide are greenhouse gases. Their increased concentration in the atmosphere is contributing to global warming. Greenhouse gases cause global warming by absorbing reflected heat from Earth’s surface thereby warming the atmosphere.
In addition to carbon dioxide, methane, and nitrous oxide, there are entirely human-made trace gases in the atmosphere that have added significantly to global warming. Halocarbons, particularly chlorofluorocarbons (CFCs) and hydro chlorofluorocarbons (HCFCs), are found in a variety of products and substances, including aerosol propellants, solvents, and refrigerants, and are released into the atmosphere from these sources. Other fluorinated gases-such as sulfur hexafluoride that has escaped from applications in electric power equipment and perfluorocarbons that have escaped from aluminum processing and the manufacture of semiconductors-are also found in trace amounts in the atmosphere.
Earth’s atmosphere is actually comprised of four layers. The troposphere starts at ground level and extends up approximately 10 mi (16 km). The next layer is the stratosphere, which is roughly 10–30 mi (16-48 km) above Earth’s surface. Above the stratosphere is the mesosphere (30–60 mi/48-97 km above Earth’s surface); and beyond the mesosphere is the thermosphere. Although each layer has a distinct composition, the troposphere and stratosphere are the layers that are critical to climate change issues.
In the troposphere, the concentration of nitrogen is around 78% and oxygen is about 21%. This is the atmospheric layer where carbon dioxide, methane, nitrous oxide, sulfur hexafluoride, and the halocarbons collect. In the stratosphere, a naturally occurring layer of ozone is formed from the reaction of sunlight with oxygen. Ozone is a form of oxygen that has three atoms in each molecule instead of the usual two atoms, and it plays an important protective role by diminishing the amount of potentially damaging ultraviolet radiation reaching Earth. CFCs and HCFCs are drifting from the troposphere into the stratosphere and destroying some of this natural ozone layer. The chlorine in the halocarbons reacts with ozone to convert molecules of ozone to oxygen, thereby depleting the ozone layer.
Also in the troposphere, carbon dioxide reacts with water vapor to make a very weak acid (carbonic acid), a reaction that depends on the temperature. Carbon dioxide gas is more soluble at colder temperatures, but saturation of the gas in water vapor is quickly reached with normal amounts of the gas in the atmosphere.
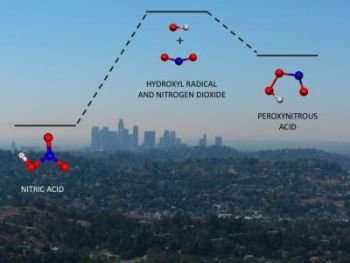
Increasing the concentration of the carbon dioxide in the atmosphere does not make the water vapor any more acidic. However, the introduction into the atmosphere of sulfur and nitrogen oxides, which react with water to form strong acids, does make the water vapor and the precipitation derived from it more acidic. This acid rain was the first recognized negative climate change that is caused by human activities.
Satellite observations have provided data on the chemical composition of the stratosphere and changes in the ozone layer. In the troposphere, no satellites are needed to observe the damaging chemical changes that have periodically occurred as a result of human activities. Photochemical smog is a serious air pollution problem that occurs when sunlight acts on nitrogen oxides, hydrocarbons, and other pollutants that are created from fossil fuel combustion. This type of smog is seen most often in highly populated areas with large numbers of vehicles during periods of high temperature and intense sunlight. In such situations, concentrations of the offending gases near ground level increase.
Impacts and Issues
Observing atmospheric chemistry requires international efforts. The World Meteorological Organization (WMO), an agency of the United Nations, has 188 participating member states and territories cooperating in a continuous study of all aspects of the atmosphere. The WMO working with the United Nations Environmental Programme established the Intergovernmental Panel on Climate Change (IPCC) in 1988 to assess information being gathered about atmospheric composition and chemistry. The IPCC is not a research organization.
It issues reports on the data collected from the WMO and other sources. In 1987, 145 countries signed the Montreal Protocol to ban the production of ozone-depleting CFCs by the year 2000. Although the use of CFCs was banned by the Montreal Protocol in 1987, the existing CFCs will remain in the atmosphere for hundreds of years. HCFCs are less damaging to the ozone layer, but their use is also being phased out because they are greenhouse gases.
In 1992, 189 countries joined the United Nations Framework Convention on Climate Change (UNFCCC) to set general goals and rules to address climate change. One result of the activities of UNFCCC was the 1997 Kyoto Protocol, which included an outline of controls for the industrial nations to contain six greenhouse gases-carbon dioxide, methane, nitrous oxide, hydrofluorocarbons (HFCs), perfluorocarbons (PFCs), and sulfur hexafluoride.
WORDS TO KNOW
Acid Rain: A form of precipitation that is significantly more acidic than neutral water, often produced as the result of industrial processes.
Fossil Fuels: Fuels formed by biological processes and transformed into solid or fluid minerals over geological time. Fossil fuels include coal, petroleum, and natural gas. Fossil fuels are non-renewable on the timescale of human civilization, because their natural replenishment would take many millions of years.
Halocarbon: Compound that contains carbon and one or more of the elements known as halons (chlorine, fluorine, or bromine). Halocarbons do not exist in nature; all are manufactured. When released into the atmosphere, many halocarbons deplete the ozone layer and have high global warming potential.
Ozone Layer: The layer of ozone that begins approximately 9.3 mi (15 km) above Earth and thins to an almost negligible amount at about 31 mi (50 km) and shields Earth from harmful ultraviolet radiation from the sun. The highest natural concentration of ozone (approximately 10 parts per million by volume) occurs in the stratosphere at approximately 15.5 mi (25 km) above Earth. The stratospheric ozone concentration changes throughout the year as stratospheric circulation changes with the seasons. Natural events such as volcanoes and solar flares can produce changes in ozone concentration, but man-made changes are of the greatest concern.
Photochemical Smog: A type of smog that forms in large cities when chemical reactions take place in the presence of sunlight; its principal component is ozone. Ozone and other oxidants are not emitted into the air directly but form from reactions involving nitrogen oxides and hydrocarbons. Because of its smog-making ability, ozone in the lower atmosphere (troposphere) is often referred to as ‘‘bad’’ozone.
Trace Gases: Gases present in Earth’s atmosphere in trace (relatively very small) amounts. All greenhouse gases happen to be trace gases, though some are more abundant than others; the most abundant greenhouse gases are CO2 (0.037%of the atmosphere) and water vapor (0.25%of the atmosphere, on average).
Ultraviolet Radiation; The energy range just beyond the violet end of the visible spectrum. Although ultraviolet radiation constitutes only about 5 percent of the total energy emitted from the sun, it is the major energy source for the stratosphere and mesosphere, playing a dominant role in both energy balance and chemical composition.
IN CONTEXT: HUMAN ACTIVITY IMPACTS NATURAL BALANCE
‘‘The current concentration of a greenhouse gas in the atmosphere is the net result of the history of its past emissions and removals from the atmosphere. The gases and aerosols considered [by the Intergovernmental Panel on Climate Change] are emitted to the atmosphere by human activities or are formed from precursor species emitted to the atmosphere. These emissions are offset by chemical and physical removal processes. With the important exception of carbon dioxide (CO2), it is generally the case that these processes remove a specific fraction of the amount of a gas in the atmosphere each year and the inverse of this removal rate gives the mean lifetime for that gas. In some cases, the removal rate may vary with gas concentration or other atmospheric properties (e.g., temperature or background chemical conditions).’’
‘‘Long-lived greenhouse gases (LLGHGs), for example, CO2, methane (CH4) and nitrous oxide (N2O), are chemically stable and persist in the atmosphere over time scales of a decade to centuries or longer, so that their emission has a long-term influence on climate.’’ ‘‘Because these gases are long lived, they become well mixed throughout the atmosphere much faster than they are removed and their global concentrations can be accurately estimated from data at a few locations. Carbon dioxide does not have a specific lifetime because it is continuously cycled between the atmosphere, oceans and land biosphere and its net removal from the atmosphere involves a range of processes with different time scales.’’
‘‘Short-lived gases (e.g., sulphur dioxide and carbon monoxide) are chemically reactive and generally removed by natural oxidation processes in the atmosphere, by removal at the surface or by washout in precipitation; their concentrations are hence highly variable.
Ozone is a significant greenhouse gas that is formed and destroyed by chemical reactions involving other species in the atmosphere. In the troposphere, the human influence on ozone occurs primarily through changes in precursor gases that lead to its formation, whereas in the stratosphere, the human influence has been primarily through changes in ozone removal rates caused by chlorofluorocarbons (CFCs) and other ozone-depleting substances.’’
Source: Solomon, S., et al. ‘‘Technical Summary.’’ In Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. New York: Cambridge University Press. 2007.
Bibliography
Web Sites
‘‘Atmosphere Changes.’’ U.S. Environmental Protection Agency. <http://www.epa.gov/climatechange/science/recentac.html> (accessed January 30, 2015).
‘‘International Global Atmospheric Chemistry Science Plan and Implementation Strategy.’’ IGBP Secretariat. <http:// http://www.noaa.gov//> (accessed January 30, 2015).
‘‘The Nobel Prize in Chemistry 1995.’’ www.Nobelprize.org
<http://nobelprize.org/nobel_prizes/chemistry/laureates/1995/index.html> (accessed January 30, 2015).
‘‘Photochemical Smog—What It Means for Us.’’ U.S. Environmental Protection Agency, March 2004. <http://www.epa.sa.gov.au/pdfs/info_photosmog.pdf> (accessed January 30, 2015).
‘‘WMO in Brief.’’ World Meteorological Organization.
<http://www.wmo.ch/pages/about/index_ en.html> (accessed January 30, 2015).