Crystallization
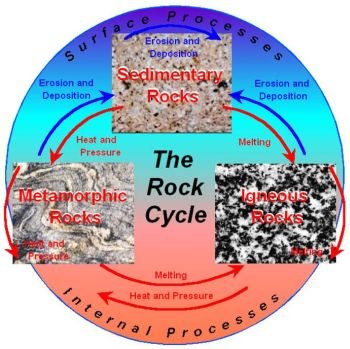
Since most magmas form in the upper mantle, their composition is mostly silica (SiO2) with different amounts of iron, calcium, sodium, potassium, aluminum, magnesium, and other trace elements. The crystallization of these compounds is known, but as the ratios of the elements are different, the crystallization looks different.
In 1912, Bowen performed a series of experiments where he compared the crystallization temperature of compounds with different silica levels. He found that minerals, which crystallized at higher temperatures (calcium-rich plagioclases, olivines, pyroxenes), were low in silica. These high-temperature minerals were further divided into a ferromagnesian class and plagioclase feldspar. The minerals that crystallized at lower temperatures were usually low in silica. In 1928, Bowen published, The Evolution of Igneous Rocks, where he focused mostly on magma. Bowen became known as the Father of Canadian Geology for his ideas on crystal formation. This mineralcrystallization cycle is known as Bowen’s Reaction Series.
The plagioclase class is a continuous reaction series where some crystals are already formed, while the rest melt. The magma composition changes continuously, but crystals that are already formed don’t change. Remember, the higher the amount of calcium in a crystal, the higher the temperature it takes to melt. As the magma cools, the crystals are constantly reacting with other elements in the melt. When cooling happens quickly, then the series shifts from high calcium-containing crystals to high silica-containing crystals.
The ferromagnesian class goes through a discontinuous reaction series. These elements begin with olivine crystallizing first, and then react with other elements in the magma melt to form pyroxene.
Mg2SiO4+SiO2+2MgSiO3
olivine+ silica in fluid magma+ pyroxene
As the magma cools and the temperature lowers even more, pyroxenes continue to react with elements in the melt. They are then converted to amphiboles. This series of reactions is interrupted (discontinuous) between each formation of different compounds. Some compounds are formed at different temperatures, before they react with the melt elements and possibly form other new compounds with different compositions.
For example, at lower temperatures, pyroxene reacts with an increase in available silica and forms amphibole. The magma continues to cool and amphibole reacts to form biotite, which contains even greater amounts of silica.
Continuous and discontinuous reaction series are separate, but the external environment also has a role. Many times, the final form of the created rock can have unique characteristics that have been brought about by the environment specifics.
Some of the environmental factors that affect crystallization are: pressure, temperature changes, rate of cooling, local deposits of calcium-or sodium rich minerals, and timing of crystallization during cooling. Magma can also be changed by the type of rock pockets that may be around it.
Magnetism
Certain minerals, the most important of which is magnetite, can become permanently magnetized. This comes about because the orbiting electrons around a nucleus form an electric current and create a very small magnetic field. A magnetic field is the space through which the force or influence of a magnet is applied.
Above a temperature called the Curie point or temperature, the thermal excitement of atoms does not allow them to become permanently magnetized. They are too busy zipping around to slow down and allow magnetism to take place.
The temperature above which all permanent magnetism is destroyed is called the Curie point or temperature.
Curie’s law, named after Pierre Curie, who with his wife, Marie, received Nobel prizes in chemistry for their work with radioactive elements. Curie’s law describes the ability of an element to be magnetized as inversely proportional to the absolute temperature. In other words, the hotter it gets, the more the atoms get excited, and the less likely magnetism is to occur.
The Curie point for magnetite is 5008C. Any temperatures higher than that cause atoms to get excited and vibrate wildly in no particular direction. This random dancing around causes the atoms’ electrical currents to cancel each other out instead of lining up and forming a stable field.
When the temperature is less than 5008C, small ‘‘islands’’ of electric current in a solid stabilize and reinforce each other. When an external magnetic field is nearby, all the magnetic islands in a solid, parallel to the magnetic field, become larger and expand, taking over the neighboring, nonparallel islands. In no time, parallel islands of current form a ‘‘continent’’ of electric current and a permanent magnet is created. This is true for cooling lava. All the minerals crystallize at temperatures above 7008C, a lot higher than the Curie points of any of the magnetic lava minerals. As crystallized lava slowly cools, its temperature drops below 5008C, the Curie point for magnetite. When this happens, all the magnetite grains in the rock turn into tiny permanent magnets. They are affected by the much greater magnetic field of the Earth.
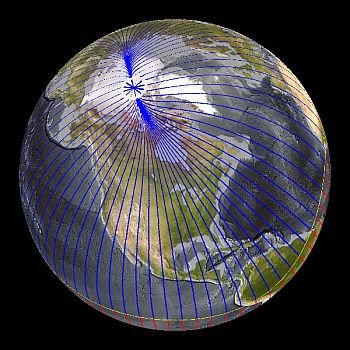
Geologists have discovered from core samples of ancient lava and modern-day lava flows that the magnetic poles of the magnetite grains in the lava sample have the same magnetic inclination as the Earth’s magnetic field.
The magnetic poles of the magnetite grains in the lava will align in the same direction as the Earth’s magnetic field. When lava samples are collected, they have unique magnetic polarities depending on the time and the magnetic field of the Earth that was in place when they were formed. The magnetic signature of lava’s formation will stay the same as long as the lava exists. The signature will be the same as when the lava’s cooling temperature passed 5008C.
This study of the magnetism of lava crystals allowed geologists to understand the changes that took place during the development and cooling of the Earth.
When the cooling of lava results in the creation of permanent magnetism, it is called thermoremanent magnetism.
No magnet is permanently magnetic. Over time, it loses magnetism. This is called the magnetic relaxation time. Permanent magnets have very long relaxation times.
Relaxation of magnets is affected by many things including the following:
- Mineral composition,
- Grain size,
- Temperature,
- Neighboring minerals, and
- The strength of the original magnetization.
The time it takes for a magnet to lose its magnetic ability is called the relaxation time.
In order for geologists to determine relaxation times and magnetization ages of rock samples, a few of the magnetic grains (perhaps those that were somewhat weaker to start with), must have already relaxed beyond the age of the first magnetization. Measuring the relaxation times of rock samples in the laboratory is performed as a temperature function.
Many igneous rock samples have relaxation times much greater than the magnetization age. These samples, collected from ancient, exposed lava flows around the world, are used to figure out where the magnetic poles were located thousands of years ago.