VIDEO
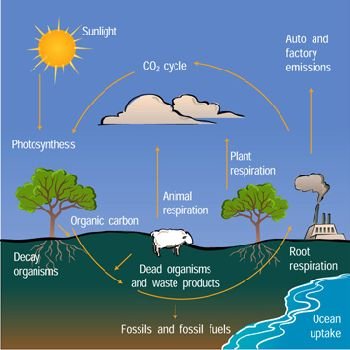
The Carbon Cycle
Understanding carbon is extremely important to understanding climate. The two most important greenhouse gases, carbon dioxide and methane, are carbon based.
Carbon only affects climate when it is in the atmosphere, but to understand the effect of carbon-based gases on climate, it is necessary to understand how these gases move through all of Earth’s major reservoirs: the atmosphere, biosphere (living things), geosphere (the solid Earth), and hydrosphere (fresh water and oceans).
The carbon cycle describes the movement of carbon between these different reservoirs. Carbon dioxide continually moves in and out of the atmosphere. CO2 leaves the atmosphere primarily through photosynthesis, the process in which plants take CO2 and water (H2O) to produce sugar (C6H12O6) and oxygen (O2).
The simplified chemical reaction for photosynthesis is:
6CO2 + 12H2O + solar energy = C6H12O6 + 6O2 + H2O
The amount of food energy created by photosynthesis is known as primary productivity. Photosynthesis is performed primarily by land plants and tiny marine plants called phytoplankton in the upper layer of the ocean. These organisms are called producers.
Photo synthesizers use CO2 from the atmosphere to build their body tissue. (Zooplankton is tiny marine animals that eat phytoplankton. Plankton refers to both phytoplankton and zooplankton.) Carbon may be stored in a single reservoir so that it is, at least temporarily, no longer part of the carbon cycle.
This is called carbon sequestration. Some important reservoirs for carbon sequestration are swamps and forests. Ancient plants and plankton are converted by earth processes into fossil fuels-oil, gas, coal, and others-which also sequester large quantities of carbon. (Currently, about 85% of primary power generation comes from fossil fuels.)
Carbon also freely enters the ocean. CO2 readily dissolves in sea-water, making the oceans into enormous carbon reservoirs. Marine organisms use CO2 from seawater to make carbonate shells and other hard parts. (A carbonate compound contains the carbonate ion CO3.
Most carbonates, including calcite and limestone, are calcium carbonates [CaCO3].) After the organisms die, some of the shells sink into the deep ocean, where they are buried by sediments. (Sediments are fragments of rocks, shells, and living organisms that range in size from dust to boulders.) This carbonate may later become part of a rock, often limestone.
The balance between the acidity of seawater and the dissolution of carbonates keeps the pH of ocean water in balance. (An acid has free hydrogen ions and can be neutralized by an alkaline substance. The measure of the balance between a solution’s acidity and its alkalinity is called its pH.) Earth processes transport some of these sediments deeper into the planet’s interior.
The carbon cycle also brings carbon back into the atmosphere. Carbon dioxide reenters the atmosphere when the processes described above are reversed, as by respiration, fire, decomposition, or volcanic eruptions. In respiration, animals and plants use oxygen to convert sugar created in photosynthesis into energy that they can use. The chemical equation for respiration looks like photosynthesis in reverse:
C6H12O6 + 6O2 = 6CO2 + 6H2O + useable energy
Note that in photosynthesis, CO2 is converted to O2, while in respiration, O2 is converted to CO2. CO2 sequestered in sediments, rock, Earth’s interior, or living things can be rereleased into the atmosphere. For example, if carbonate rock is exposed to the atmosphere, the rock weathers and releases in Earth are interior and inject it into the atmosphere.
Forests lose carbon to the atmosphere if they decompose or are burned. CO2 is rereleased into the atmosphere when fossil fuels are burned. Scientists estimate that recoverable fossil fuel reserves contain about five times as much carbon as is currently in the atmosphere. Water temperature affects the ability of the oceans to store carbon. Cold water holds more gas, so cold seawater absorbs CO2 from the atmosphere. Conversely, gases bubble up as seawater warms and reenters the atmosphere.
Like carbon dioxide, methane enters the atmosphere in a variety of ways. Methane forms primarily as single-celled bacteria and other organisms break down organic substances sewage, plant material, or food-in the absence of oxygen. Methane enters the atmosphere during volcanic eruptions and from mud volcanoes. Methane is the primary component of natural gas, which forms in a process that is similar to the process that forms other fossil fuels.
Natural gas formation removes methane from the atmosphere. The methane is rereleased into the atmosphere when natural gas is burned. The atmosphere also loses methane when CH4 undergoes a reaction with hydroxyl (OH) ions. Over time, atmospheric methane breaks down to form CO2. Living plants may also add methane to the atmosphere, although scientists are just beginning to explore this idea. Methane is found in offshore sediments in enormous quantities as methane hydrates.
These compounds develop at depths of 660 to 1,650 feet (200 to 500 m) below sea level when decomposed organic matter contacts cold water at the high pressures found deep in layered sediments. Water molecules form an icy cage (a hydrate) that contains a methane molecule. The molecule’s structure is unstable; when the pressure is removed from the hydrate, the structure collapses, and the methane escapes. Methane hydrates can also be used as fuel, although the technology for mining them and harnessing their energy has not yet been developed. Thousands of gigatons of methane, equal to the world’s total amount of coal, are located in the oceans.
WrapUp
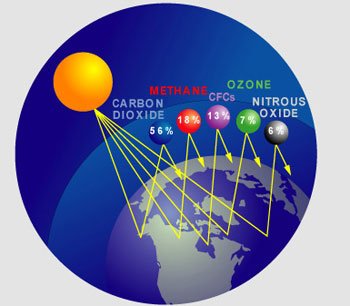
Earth’s climate is a complex system. In any location, climate is determined by latitude, proximity to an ocean, and position relative to atmospheric and oceanic currents, altitude and albedo, plus other factors. One of the most important determinants of Earth’s global climate is atmospheric greenhouse gases. Because greenhouse gases trap some of the heat that radiates from Earth’s surface, an increase in their abundance causes global warming, the ongoing rise in average global temperatures. Due to their abundance, the carbon-based gases carbon dioxide and methane are the most important greenhouse gases.
Carbon cycles in and out of the atmosphere: It is sequestered in various reservoirs, such as fossil fuels and trees, but it is also released back into the atmosphere when, for example, those commodities are burned. Small changes in any of the features that regulate climate may cause the climate to change locally or globally.