A Climate Minute – The Greenhouse Effect
Origin and Impact of Greenhouse Gases
The climate of the earth has gone through a recurring pattern of temperature changes which has brought the earth in and out of a series of historic ice ages. Gases in the atmosphere also have ebbed and fl owed with the temperature cycles. However, the levels of greenhouse gases building up in the atmosphere today have never been this high throughout Earth’s history.
What People Add to Nature’s Greenhouse?
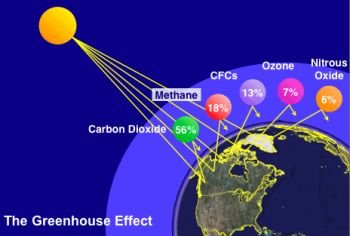
Each year human activities increase the concentrations of the greenhouse gases in the atmosphere. The concentrations of these gases have increased each decade. Carbon dioxide from the burning of fossil fuels stands out in terms of both being the largest greenhouse gas component and also increasing at the fastest rate.
CARBON DIOXIDE
Carbon dioxide is increasing in the atmosphere. Its concentration is now over 380 parts per million (ppm) and rising rapidly. At no time in this planet’s history has the carbon dioxide level ever been above 300 ppm. Today, it is at levels that are unprecedented in recent history, and it is growing at a rate that, if it continues, is likely to have a major impact on global climate.
Each year nature puts about 200 billion tons of carbon dioxide into the atmosphere, mostly from decaying plants and spewing from volcanoes. This is 30 times more than what we release into our atmosphere from our cars, power plants, and factories. However, the hundred billion or so tons of carbon dioxide that we have added to the atmosphere since 1850 are enough to initiate the climate changes that we are beginning to witness around the world. Since we add roughly 7 billion tons each year at current rates, we are continuing to tip the balance.
Nature produces and absorbs carbon dioxide, establishing what is called the preindustrial level of about 280 ppm in the air. This means that for every million air molecules, 280 of them are carbon dioxide. Today, the global carbon dioxide level, as a result of human activities, is nearly 100 ppm higher than the natural level. At current rates, this may reach a level of 560 ppm by the end of this century.
Contributions from humans are known as anthropogenic greenhouse gases. Carbon dioxide is at the top of the list because of its impact on global warming. Burning fossil fuels primarily for electricity generation, transportation, industrial process heat, and heating buildings contributes carbon dioxide. Cement production stands. Forests absorb carbon dioxide through the action of photosynthesis and are considered a natural carbon sink because they remove carbon dioxide from the atmosphere. Beginning in the mid-1700s, settlers in North America began to clear forest land to make room for the massive westward migration that was about to take place. As the settlers removed trees, they also were removing the natural sinks, and carbon dioxide levels started climbing.
Carbon dioxide emissions are measured in gigatons of carbon dioxide (GtCO2). The prefix giga means “billion.” Between the years 2000 and 2005, 7.2 GtCO2 (7.2 billion tons of carbon dioxide) was added each year to the atmosphere. This is an increase above the 6.4 GtCO2 emissions each year from 1900 to 1999. This represents an increase of 12.5 percent from one decade to the next.
About half the carbon dioxide that humans add to the atmosphere is removed by nature, primarily by the oceans. The more saturated the oceans become, the less they are able to continue taking carbon dioxide out of the atmosphere.
Carbon dioxide emissions come mainly from the combustion of fossil fuels. This occurs mostly in industrialized areas. Main areas of concentration are North America, Europe, and Southeast Asia. These sites do not include the contributions of mobile sources such as cars.
Compare this with a view from space provided by the NASA Aqua spacecraft. Carbon dioxide (which in this case also includes contributions from moving sources) hovers around the urban and industrial centers. Lower carbon dioxide concentrations are found over the oceans and poles.
Some mixing of gases may have occurred in the atmosphere, but the highest concentrations of carbon dioxide are centered closest to their sources.
MEASURING GREENHOUSE GAS EMISSIONS
Scientists use several different ways to quantify how much of a greenhouse gas is being pumped into the atmosphere over a given time period. This is important in formulating a solution to the problem. It is also important when organizations may be assessed penalties or given credits for their greenhouse emissions.
Here are three different methods for keeping track:
1. Gigatons of carbon dioxide (GtCO2). This is a straightforward measure of how many (billions of) tons of carbon dioxide are emitted by a site.
2. Gigatons of carbon (GtC). Some people like to talk about “carbon” emissions or “carbon footprints.” GtC measures the same thing as GtCO2 but focuses only on the weight of the carbon, not the carbon dioxide. This number is 27.3 percent smaller than GtCO2, reflecting the percentage of carbon in carbon dioxide by weight.
3. Carbon dioxide equivalents (GtCO2, eq). Since there are a number of greenhouse gases, to avoid dealing with each one separately, a single index is used to measure the overall effect of all the greenhouse gases having an equivalent effect as carbon dioxide. Thus, if carbon dioxide, methane, and nitrous oxide are being generated in a certain region, one number can sum up the entire impact. This is called the carbon dioxide equivalent.
Note: Occasionally, greenhouse emissions may be reported in kilograms or in tones, which is a 1000-kg unit. It is important to compare apples with apples.
METHANE
The primary sources of methane in the atmosphere are agriculture and the use of fossil fuels. The main natural sources of methane are from decomposition of organic matter in wetlands, rice paddies, and bogs. Termites are also a source of methane.
Methane was once called “swamp gas” and “marsh gas” because it was commonly observed to evolve from wetland areas filled with decaying organic material such as leaves and other vegetation.
Methane is a by-product of the digestion of farm animals such as cows and pigs (in a process called enteric fermentation). The amount of methane depends on the diet of the animals and how the manure they produce is handled. The tendency toward larger farms favors production methods that release more methane, although the opportunity is also greater to contain it.
Methane is a main component of natural gas (the other component being ethane). Leaky natural gas pipelines inadvertently release methane, which is also released during the process of extracting natural gas or petroleum from the ground.
One molecule of methane absorbs 20 times the amount of sun energy of a molecule of carbon dioxide. However, methane is now at a level of 1774 parts per billion (ppb) in the atmosphere, which is 165 times less prevalent than carbon dioxide. (As Austin Powers might say, this is parts per billion, not millions, which in the case of concentration is much less.) For this reason, methane has less than a third of the overall impact on climate that carbon dioxide has. Studies of the relative proportion of carbon isotopes in the atmosphere indicate that the human-contributed level of methane is well above the natural level. Methane has increased from fossil fuel use, but emissions from agriculture have been stable. In contrast to carbon dioxide, the growth rate of methane emissions overall actually has declined since the 1990s. The main methane sink is a photochemical process that removes methane from the atmosphere. The molecules involved are water vapor, ozone, and oxygen interacting in the presence of light to destroy the methane molecule. Together they form a piece of a molecule called a hydroxyl radical (OH–) that reacts with the methane and whose presence in the atmosphere serves as an indicator of how long methane will persist. The average lifetime for methane in the atmosphere is about 12 years, which is much less than carbon dioxide.
Methane clathrate is a slushy semi frozen mix of methane gas and ice typically found in the northern hemisphere’s tundra permafrost regions and in sediment layers on the ocean floor. Some scientists have speculated that a sudden release of large amounts of methane from deposited methane clathrate called the, clathrate gun theory, might be a cause of abrupt past and possibly future climate changes.
However, one study of ice core samples from Greenland performed by Dr. E. Brook of Oregon State University did not find a sudden increase in methane levels in past atmospheres that might have accounted for precipitous historic temperature increases.
NITROUS OXIDE
More than a third of the nitrous oxide put in the atmosphere by humans comes from the use of nitrogen-based fertilizers in agriculture. Nitrous oxide is the well-known dental anesthetic known as “laughing gas.” Some nitrous oxide is formed as a result of biomass burning and certain industrial processes such as nylon production.
The main sink for nitrous oxide is a photochemical process in the stratosphere involving oxygen and resulting in a relatively long lifetime in the atmosphere of about 115–120 years. Nitrous oxide, like the other greenhouse gases, has been increasing steadily above its preindustrial values.
CHLOROFLUOROCARBONS (CFCs) AND OZONE
Ultraviolet Protection
CFCs are human-made chemicals that do not exist in nature. Since they become a vapor just below room temperature and are nonflammable and nontoxic, they were used widely in refrigeration systems, insulation, and as a propellant in spray cans in the 1980s. Because they are highly stable chemically, once released into the atmosphere, they can persist for a few hundred years. Like an unwelcome house guest, they are released into the atmosphere and are pretty much there for good.
All CFCs as a group have a concentration in the atmosphere of around 1 ppb. This may seem like an insignificant presence, but it was enough to destroy ozone in the stratosphere. Stratospheric ozone protects the inhabitants of the earth by absorbing ultraviolet rays from the sun before they can enter the atmosphere. Stratospheric ozone is like nature’s sun block.
In response to concerns about ozone depletion, many governments signed the Montreal Protocol in 1989, which required the phasing out of CFCs entirely. The industrial countries were given until 1996 to do this, and the developing countries had until 2006. In terms of international cooperation, this is a success story. As a result of implementation of the Montreal Protocol, CFCs are no longer increasing in the atmosphere. Because of the very long lifetime of CFCs in the atmosphere, though, they are expected to be present near current levels for hundreds of years.
CFCs and Ozone as Greenhouse Gases
In terms of global warming, both ozone and CFCs are greenhouse gases. The presence of CFCs is partly compensated by their tendency to destroy ozone. The CFC molecules make up for their relatively small abundance by being virtual “infrared sponges,” absorbing 5000–10,000 times the amount of energy from the sun as a carbon dioxide molecule. On balance, they contribute about 20 percent of the overall atmospheric heating owing to the current mix of greenhouse gases. The CFCs were replaced by a class of chemicals called halocarbons. (The chemical prefix halo refers to the halogens, which include the elements fluorine, chlorine, bromine, and iodine.). These include hydrochlorofluorocarbons (HCFCs) and hydrofluorocarbons (HFCs). The HCFCs are less destructive to ozone and are preferable for that reason. Both, however, are greenhouse gases, although they have shorter lifetimes on the order of decades rather than centuries. HCFCs are slated for being phased out by 2030.
Other compounds in this group are the perfluourocarbons (CF4 and C2F6) and sulfur hexafluoride (SF6), which are used in industrial processes. Although they are present in the atmosphere in small quantities, they have 1000-year lifetimes. Ozone in the stratosphere absorbs ultraviolet light, preventing it from being absorbed closer to the earth’s surface. Some of this can be readily radiated back into space. Thus ozone in the stratosphere actually has a slight cooling effect on the earth’s overall energy balance.
AEROSOLS-AN UMBRELLA ABOVE THE GREENHOUSE
Human activities also add solid suspended material-called aerosols-to the atmosphere. These include suspended carbon (soot and ash), carbon in compounds, dust, and oxides of sulfur and nitrogen. Together, the aerosols produce a cooling effect by reflecting incoming sunlight back into space before it has a chance to enter the atmosphere. Overall, the aerosols act to restrict some of the sunlight from heating the earth. In some cases they absorb some of the incoming light. Aerosols also can influence cloud formation and precipitation





